The 7 wonders of the cellular world
A cell biologist's tier list
Foreword: This article dives into some cell biology—a first for my Substack. Richard Feynman said, “If you can’t explain something in simple terms, you don’t understand it”. Having done my PhD in cell biology and immunology, I should understand this stuff, so I should be able to explain it in simple terms! Hopefully I can convey some of the awe and intrigue that keeps us cell biologists going, whilst keeping it pretty accessible. As always, I’ve recorded a voiceover for this one, but I’d encourage reading it so you get the full benefit of the pictures and videos. If you enjoy, please let me know by way of clicking the various clickable things. Thanks! - Adam
Introduction
Cells are the smallest indivisible units of biology. In my biased view, every biological process and every disease is best understood through the lens of cell biology. Seemingly macroscopic physiological phenomena, like day-night rhythms in our behaviour, ultimately boil down to cells (circadian rhythms are maintained by autonomous molecular “clocks” that run indefinitely in each of our cells). At the other end of the spectrum, atomic or molecular processes are devoid of biological importance until incorporated into a cellular process. For example, Franklin’s famous X-ray crystallograph (“photo 51”) was only useful and interesting to Watson and Crick because they were trying to understand how cells store and read information.
I think understanding how cells work is really important. And really interesting. Here, I want to give an overview of what I consider to be the seven wonders of the cellular world. These are all macromolecular structures inside of cells that each play a key role in sustaining the cell, and, by extension, life. Of course, organelles can be described as wonders of the cellular world (e.g., the Golgi Apparatus or the mitochondrion) but I thought sticking with large molecular complexes keeps the game fairer. Hard to compete with the powerhouse of the cell itself! Factors that come into play for making this list include aesthetic beauty, mysterious aura, remarkable function, and essentiality for life. If you’re not a scientist, this list will hopefully give a broad intro into cell biology, with each of my seven picks taking you on a journey through the cell both spatially and functionally. I say all seven picks, but I mean all six since the last one really is a complete mystery even 50 years on from its discovery. With that in mind, let’s learn some cell biology!
1. The nucleosome
Cells synthesise two main types of complex biopolymers: nucleic acids (like DNA and RNA) and polypeptides (which form proteins). Watson and Crick’s discovery1 of the structure of DNA helped to lay the groundwork for Crick’s later description of the “central dogma of molecular biology”. This key concept basically states that information in DNA can be copied into RNA and, in turn, into protein. But information in proteins cannot go back into nucleic acids (DNA/RNA). Information flows this way because DNA is primarily a stable source of genetic information, and proteins (long chains of amino acids folded into complex 3D structures) are the functional units within cells—they’re the things that actually do useful stuff. Cells transcribe DNA into shorter chunks of RNA, which mostly act as intermediate messengers, and this RNA then gets translated into protein.
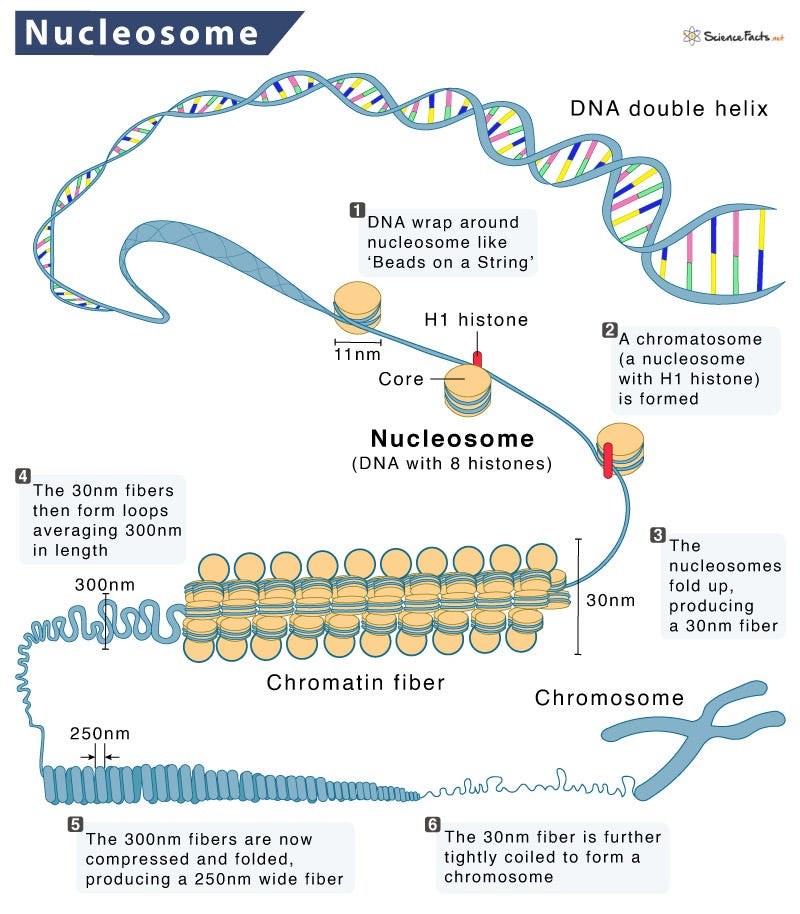
The genome is the total DNA code needed to produce every single type of protein that any cell within an organism could need (and much more non-protein stuff, actually). This is a lot of information—for humans it’s about 6.4 billion bits of information in 3.2 billion base pairs, or “letters” of DNA. If the chromosomes were unravelled and joined end-on-end, each human cell would have about 2 metres of DNA. So how the hell does the cell stuff this all into the nucleus, which is about 5 micrometers in diameter at most? Enter: the nucleosome.

Discovered in 1974 by married couple Don and Ada Olins (who sign off letters and emails with “DnA”), the nucleosome is formed by proteins called histones stacked into a cylinder with DNA wrapping around the outside in a coil. If the nucleus of a cell is burst open with pure water and the contents imaged by an electron microscope, you can see nucleosomes as “beads on a string”, as in the image above. The nucleosome is the most fundamental storage unit of DNA in cells and is essential for large genomes and, therefore, complex life.
The insane level of spatial compression needed to cram the two copies of the genome into every one of our cells doesn’t solely rest on the shoulders of the nucleosome though. These coils of DNA themselves form coiled stacks, called chromatin fibres, which in turn are further coiled, compressed, and folded. Chromatin, which describes the overall organisation of DNA in the cell, is not merely static though. The organisation of these nucleosomes is dynamically regulated to control which genes can get turned on or off in a cell. Opening up the chromatin allows greater access to the naked DNA not coiled up in the nucleosome, which allows that DNA to be read and copied into RNA. It’s basically like those archives or libraries with sliding shelves that students try to through before they close. If you want to take out a particular book (in this analogy: a gene), then you open up the channel between the shelves where it is located (in this analogy: chromatin opening up for transcription).
Given their crucial role in affording cells their incredible information density, as well as their simple but beautiful beads-on-a-string arrangement, nucleosomes gain a spot on this list.
2. The nuclear pore complex
We’ve covered how DNA, which is kept in the nucleus, is copied in short chunks into RNA, which can then be used as the template from which to make proteins with useful functions. These proteins don’t get made in the nucleus though—it’s way too crowded. Further, the nucleus is basically a fortress. It’s encapsulated with a thick double membrane so that stuff that can damage or interact with DNA in undesirable ways is kept out, and the DNA itself is kept in. If any DNA were to get out, the cell would think it’s under attack from a virus (which is the only other scenario where you get DNA in the cytosol—the soup of stuff outside of the nucleus), and the cell would probably kill itself. So the RNA gets made inside the nucleus but then must leave and get into the cytoplasm where proteins are made. The gateways between the nucleus and the rest of the cell come in the form of many nuclear pores. Far from just holes in the nuclear membrane though, these are stringently regulated by the nuclear pore complex.
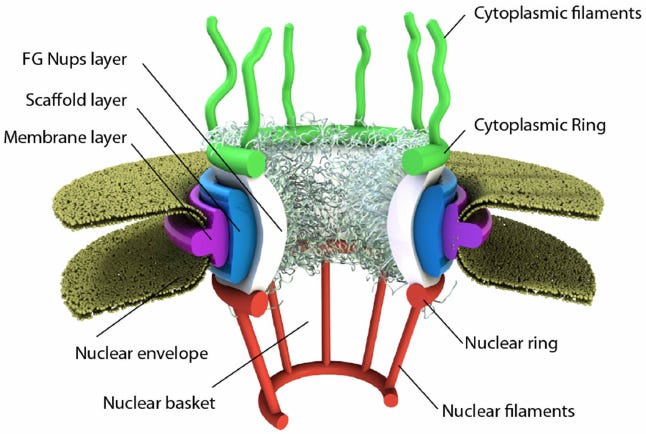
Made up of a bunch of different individual proteins called nucleoporins, the nuclear pore complex is a massive and complicated machine. It sits in the nuclear membrane and forms a pore in its centre which allows small molecules to freely diffuse, but bars anything of size and functional significance from passing. For these molecules to pass through, like big proteins trying to get in or freshly transcribed RNA molecules trying to get out, they have to be tagged by a molecular chaperone that escorts them through and out the other side.
We still don’t know exactly what it looks like at the atomic level, mainly because the very flexible bits on either side are hard to resolve via crystallography or electron microscopy, but some solid efforts have elucidated the beautiful symmetry of the core ring structure, which it turns out is dilatable. Massive geometrically satisfying nightclub bouncers, these things are easily a wonder of the cellular world.
3. The ribosome
Recap: we have DNA in the nucleus, and we know that this gets copied into RNA which can then leave the nucleus through the nuclear pores, and this then forms the template for making proteins. As I’ve mentioned, cells are basically protein machines, spatially organised into lipid-membraned compartments, termed “organelles”. Proteins are biomolecules comprised of chains of amino acids that form unique 3D structures that give them functional properties. Haemoglobin is a protein than can bind to oxygen for its transport around the body. Collagen is a protein that confers tremendous tensile strength that is useful in tendons and skin. Histones and nucleoporins are proteins that you are already an expert on. So how does the cell make all of these proteins? That’s where the ribosome comes into play.
Ribosomes are massive ribonucleoproteins, meaning they themselves are made not just of protein, but also RNA—specifically ribosomal RNA, or rRNA. They assemble around a messenger RNA (mRNA) molecule, produced when the cell reads a gene in your DNA and wants to get the “message” out of the nucleus. Ribosomes loaded onto an mRNA then process along the genetic code, taking in matching amino acids for every codon of genetic message, and stitch them together to form a long polypeptide chain that then folds to become a mature protein. Since the ribosome converts the language of nucleic acids into the language of proteins, using triplets of RNA letters called “codons” to encode one of the 20 available amino acids, this process is called translation.

Since producing proteins is such a key feature of cells, these machines evolved a very very long time ago. This makes ribosomes in different organisms all pretty similar, all things considered. As such, rRNA is the go-to target of sequencing for microbiome research, since every type of bacterium in your gut microbiome will have a recognisable rRNA, making them a good candidate for comparing different bacterial species side by side. rRNA is also a really useful yardstick to help with quality control when sequencing the mRNAs inside a cell, a.k.a. “transcriptomics”—something biologists bloody love doing these days.
Each of our cells has tens of millions of these bulky molecular machines relentlessly chugging along at about 4 amino acids per second. These masterpieces of molecular evolution allowed proteins to first become a thing for life on earth—since the first semblances of life on earth probably started with just RNA. Next time you’re in the gym and notice that your muscles have gotten a little bit bigger, be sure to give a quick thanks to your ribosomes.
4. Microtubules

Once proteins are produced, they have to get to the right place within the cell. The cell has many ways of moving things around to where they need to be. Perhaps the fastest and grandest, though, is via microtubules. The highways of the cell, these are long hollow tubes formed of two protein subunits called alpha-tubulin and beta-tubulin. These dimerise and then stack up in a spiral formation to form tubes with pseudo-helical symmetry comprised of 13 proto-filaments (see image below to make sense of that). Microtubules can polymerise and also undergo “catastrophe” whereby they fall apart from one end. This allows them to grow and shrink, allowing the cell to regulate them dynamically.
Microtubules form “train tracks” that motor proteins like dynein and kinesins use to “walk” along, dragging cargo as they do. If the cell needs to transport something quickly and a long distance, it goes via the microtubules. Long distance high-speed transport is key for many cell types, but perhaps most especially neurons, which have long extensions from the main cell body to the neuronal synapse. These extensions, called axons, contain bundles of parallel microtubules that are crucial for the transport of lipid vesicles, mitochondria, and even mRNA. My favourite cell type, cytotoxic T lymphocytes (a.k.a. “killer T cells”), point one end of all of their microtubules at cancer cells that they engage with, and use the highways to deliver cytotoxic proteins to these cancer cells to kill them.
A quirky feature of the tubulin subunits of microtubules is that they have a lot of a certain type of amino acid in them that allow electrons to become delocalised. Because of the highly ordered arrangement of subunits, the region of delocalisation is very large. This gives microtubules the ability to harness quantum effects at larger scales — effects such as superradiance. This, combined with the fact that microtubules are really abundant and important in the brain, and many anaesthetics seem to work by targeting microtubules specifically, gives rise to the notion that microtubules might be the biological basis for consciousness and free will (a hypothesis that I discuss in greater depth in my essay on why I believe free will exists). Interestingly, one of the key proteins that seems to aggregate in Alzheimer’s disease is a microtubule-interacting protein.
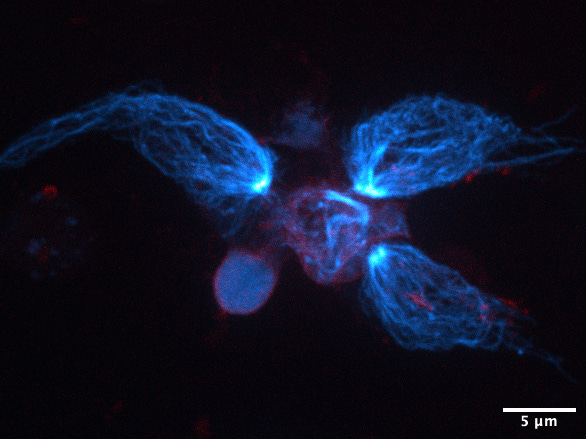
These incredible structures were one of my favourite things to image under the microscope during my PhD. Their structure is beautiful at every scale, they have vital and diverse roles in different cell types, and they are still shrouded in plenty of mystery (what is going on in their hollow cores?!). All of this earns them a solid spot as one of the 7 wonders of the cellular world.
5. The proteasome
We’ve spoken about how the cell needs to churn out a lot of proteins to achieve its functions. But just creating proteins makes for a very messy soup. The cell also needs a way to deliberately get rid of unwanted proteins. Autophagy is one way of doing this, and it allows the cell to recycle lots of proteins to create new amino acids when cells are starved of nutrients. But autophagy is typically a cell-survival mechanism, and can be indiscriminate regarding the proteins that it targets. To target specific proteins for degradation with high precision to keep things running in perfect balance, the cell uses proteasomes.
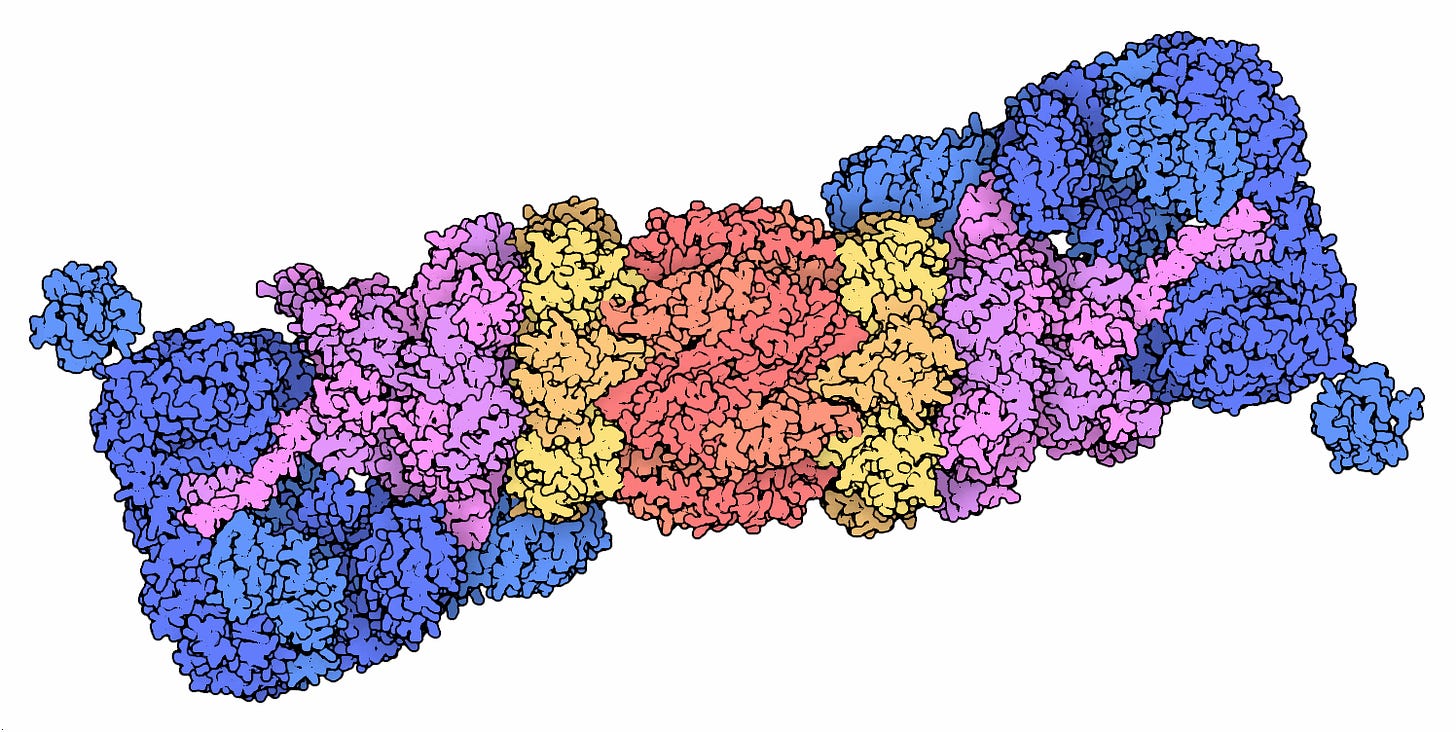
The proteasome is a very large hollow cylindrical protein complex that targets proteins that have been tagged for destruction via a little molecular red flag, known as ubiquitin. Ubiquitinated proteins are unfolded and fed into the core of the proteasome by the cap structures (magenta in the image above). The core then has a series of proteases (enzymes that chop up proteins) on the inside, using ATP to provide energy to break up the peptide chemical bonds. The hollow structure of the proteasome prevents these proteases from otherwise wreaking havoc on the rest of the cell’s proteins.
Recent work is uncovering how protein sequences, called degrons, lead to certain proteins getting destroyed. In some cancers, these sequences are mutated, meaning proteins that would otherwise get chopped up by the proteasome don’t, and this can lead to cancer. Leveraging degrons to get cells to destroy cancerous or otherwise pathogenic proteins is a promising area of future research. The garbage disposal of the cell. Vital, beautiful, and with great biotechnological promise—proteasomes are a wonder of the cellular world.
6. ATP synthase
To fuel nucleosome assembly, nuclear pore entry/exit, ribosomes, microtubular transport, proteasomes, and most other active cellular processes, the cell uses adenosine triphosphate, or ATP. This little chemical is a great currency for energy, since the third phosphate group can be chemically reacted on and off, either requiring or generating a packet of chemical energy. When proteins use ATP, they cleave that third phosphate and get energy as a byproduct of the reaction. As such, the cell needs to constantly upkeep its ATP stores to make sure there is enough of it diffusing around that proteins can use it all of the time.
This is where ATP synthase comes in. Located in the inner membrane of mitochondria, these molecular turbines are remarkable machines that add the third phosphate to ADP molecules. As you might have guessed, this requires energy. This energy comes in the form of a gradient of hydrogen ions, or protons, which mitochondria maintain across this inner membrane. In fact, the whole point of mitochondria is to establish this gradient of proton concentration. The mitochondria have two membranes. They pump loads of protons into the gap between these membranes, using the chemical energy from our food and oxygen.
The ATP synthase molecule sits in the inner membrane, which is impermeable to ions. There are gaps in between subunits of ATP synthase that allow protons to slide in, attempting to get across the membrane. As they do so, the whole machine rotates like a turbine. This rotating motion is then used elsewhere in the protein complex to move atoms around to whack on the third phosphate group to an ADP to generate ATP.
It’s truly insane. A literal molecular turbine. The analogy of hydroelectric electricity is a good one. Getting the water up there is done by clouds and rain etc, but in the cell its the mitochondria doing that work. The hydroelectric power plant is the ATP synthase. Hydrogen ions flow “downhill” from high to low concentration and spin the turbine, which whizzes away at around 250 revolutions per second (significantly faster than the average jet engine at around 150 revolutions per second).
The dude who figured out the structure and mechanism of this beast won the Nobel prize the year I was born, and he still works in the building I did my PhD in. Despite being as old as me, there is still some mystery surrounding ATP synthase. We don’t exactly know what the outside jacket of the turbine looks like, which is kind of important for knowing exactly how the hydrogen ions generate such phenomenal RPMs.
This marvel just has to be visualised to be fully appreciated, and I’d highly encourage you to watch the embedded video below when you get five spare minutes (perhaps after reading about the 7th wonder below …).
7. The vault
Finally, we come to the most mysterious wonder of them all. In the 1980s, researchers were trying to isolate different kinds of vesicles (lipid-encapsulated little droplets inside cells). They noticed these little specks in their electron microscopy samples. Around triple the size of ribosomes, which held the title for largest protein complex at the time, they assumed these specks were just impurities. Turns out, they were their own thing entirely. Once isolated and imaged properly under an electron microscope, their structure looked a bit like the vaulted ceilings of cathedrals, and thus they were named vaults.

Vaults are huge, beautifully symmetrical, hollow structures. The outside is made of a single protein, called major vault protein (MVP), of which there are 39 copies per half. Mammalian cells have loads of these things—hundreds of thousands up to millions in certain immune cells. But at the same time, plants, fungi, and bacteria lack them. The fact that single celled amoebae have them but things like insects don’t implies that they evolved a long time ago but were since lost by the multicellular organisms that today lack them.

If they’re so ancient and so massive (making them expensive for the cell to make), and so similar across the organisms that have them, then they must be important right? But if you get rid of them in mice, the mice are completely unaffected as far as we can tell, which implies no function at all …
We do have some hints to their function, but no coherent hypothesis satisfies all of the evidence. They tend to cluster around the nuclear pores, so they could be little transport pods for the cell. Their natural cargo seems to be unique RNA species, terms vRNAs, but what these do also isn’t clear exactly. There is some evidence they they help fight bacterial infections in the lung epithelium. Alternatively, given there are many more of them in macrophages, immune cells that migrate a lot, and also single-celled slime moulds which also migrate a lot, perhaps they have a role in cellular locomotion?
The guy who co-wrote the textbook on cell biology thinks vaults “may be part of the massive war with viruses, enabling cells and organisms to survive a type of virus that we don’t even yet know anything about”. While he can’t hide his fascination, he still refuses to put them in his textbook at all—I only found out about these things half way through my PhD!
The enigma endures, and scientists are still trying to solve the mystery. Some super recent evidence from May this year suggests that their movement up and down neurons might be important for learning and memory, but clearly this can’t be a universal function since theyre found in many cells, including single-celled organisms.
Ylvis are kept up at night by Stonehenge. Cell biology PhD students are kept up at night by the vault.


awesome writing style, awesome visuals